|
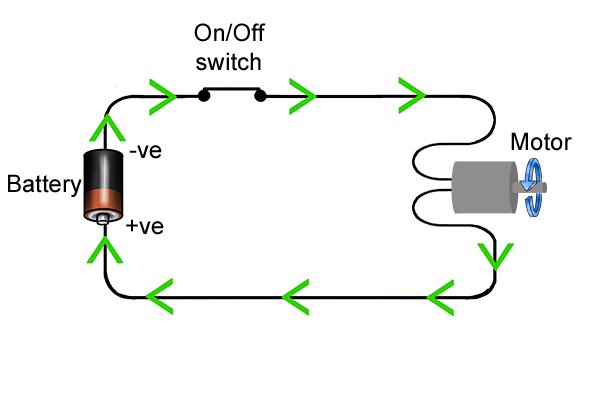
 | Amabhethri asebenza ngokugcina amandla futhi awakhulule lapho ukujikeleza kukagesi kuqediwe. Amandla angasetshenziswa futhi asetshenziswe ukudala ukukhanya, ukushisa, noma ukunyakaza. La mandla avame ukubizwa ngokuthi ugesi. |
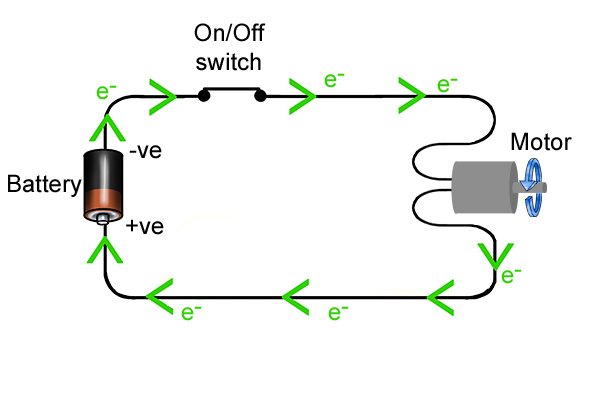
 | Uma ucindezela inkinobho yamandla ethuluzini lamandla angenazintambo, uqedela ukujikeleza kagesi okuvumela amandla ukuthi ageleze esuka kubhethri aye ethuluzini futhi enze i-drill ijikeleze, isibonelo. |
 | Ibhethri lingagcina inani elilinganiselwe lamandla kuphela, futhi lapho liphela, lidinga ukushajwa kabusha ngeshaja. Ishaja isebenzisa ugesi ophuma kumapayipi amakhulu ukuze igcwalise ibhethri ngamandla, futhi isilungele ukusetshenziswa futhi. |
 | Uma unentshisekelo kukhemistri eyenza konke kusebenze, qhubeka funda! |
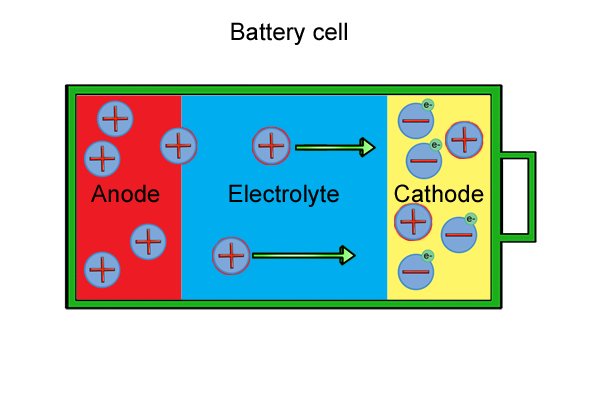
ikhemikhali yebhethri |
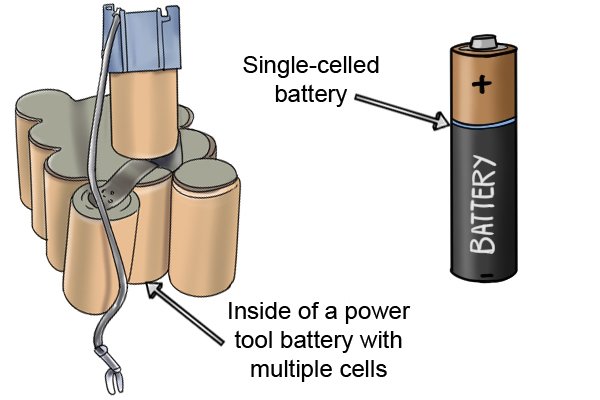
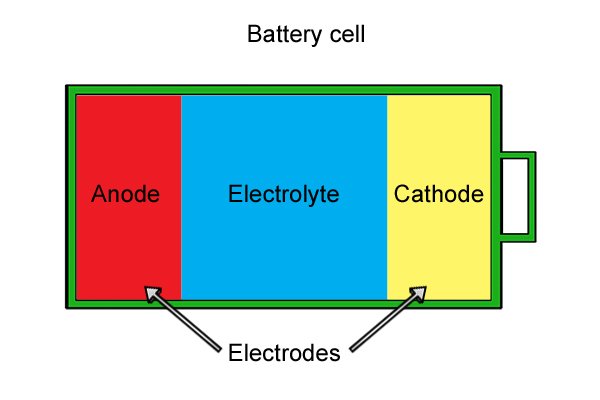
 | Ibhethri elidizayinelwe amathuluzi wamandla angenantambo lakhiwe "amaseli" ambalwa ebhethri futhi laziwa njengephakethe lebhethri. Uma amaseli engeziwe, umsebenzi omningi ibhethri elingakwazi ukuwenza ngaphambi kokuthi liphele. |
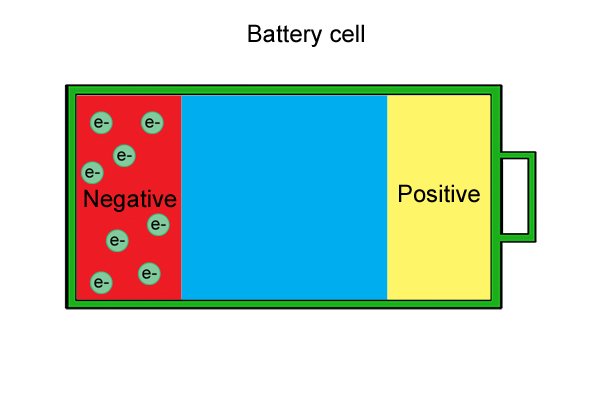
 | Ngaphakathi kweseli ngalinye kune-anode, i-cathode kanye ne-electrolyte. I-anode ne-cathode, ngokuhlangene eyaziwa ngokuthi "ama-electrodes," zenziwe ngezinto ezisabelayo lapho zihlanganiswa. I-electrolyte iwuketshezi noma unamathisele omanzi ohlukanisa ama-electrode komunye nomunye. |
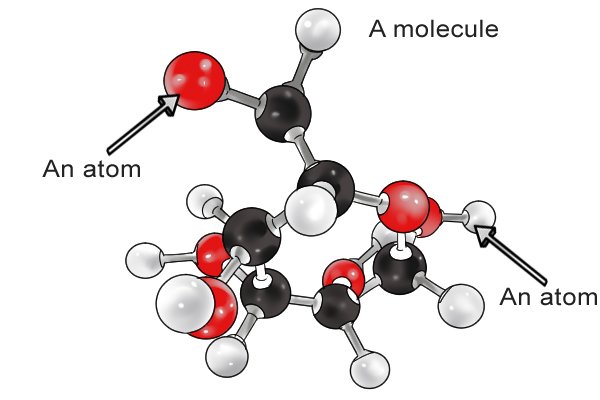
 | Yonke into emhlabeni yakhiwe ama- molecule amancane asebenzisanayo ngokusekelwe eshadini yawo kagesi (enhle, engalungile, noma engathathi hlangothi). Ukuze siqonde ibhethri, kudingeka sibheke ukuthi ama-molecule aku-electrode axhumana kanjani. |
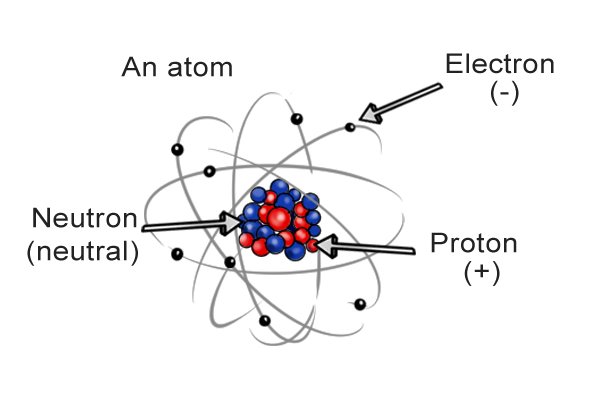
 | I-molecule yakhiwe i-athomu eyodwa noma ngaphezulu, okungamabhulokhi wokwakha amancane kakhulu. |
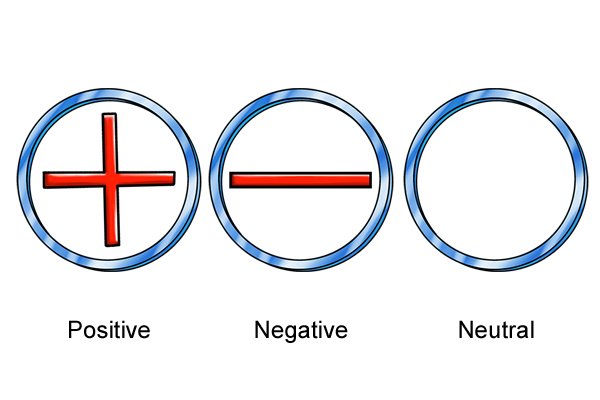
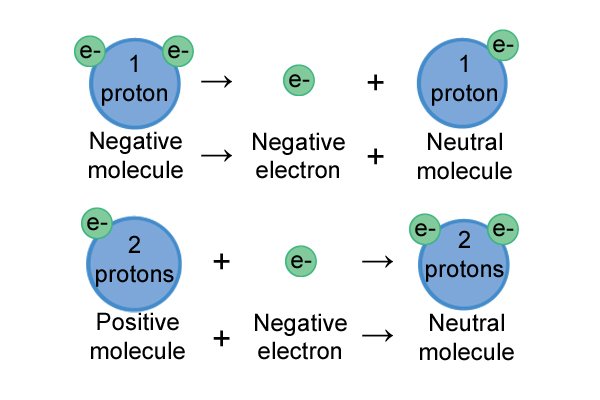
 | I-athomu ngayinye ine "nucleus" phakathi nendawo equkethe ama-neutron nama-proton. Ama-electron azungeza i-nucleus. Ama-neutron awathathi hlangothi, ama-proton alungile, nama-electron anegethivu. Ibhalansi phakathi kwamashaji inquma inani lokushajwa kwe-athomu, futhi ibhalansi phakathi kwama-athomu ku-molecule inquma inani lokushaja kwe-molecule. |
 | Yonke i-molecule ifuna ukungathathi hlangothi. Okuwukuphela kwendlela abangakwenza ngayo lokhu ukulahlekelwa noma ukuzuza ama-electron. Uma babelana ngenkokhelo ephozithivu, baheha ama-electron; uma babelana ngeshaji elibi, balahlekelwa ama-electron. |
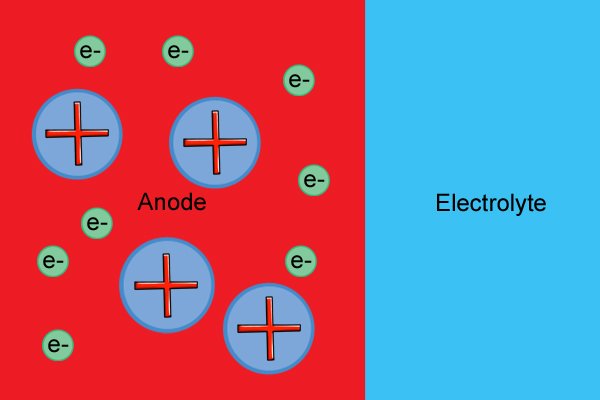
 | Ama-molecule e-anode awathathi hlangothi kuze kube yilapho esabela nge-electrolyte, ebangela ukukhululwa kwama-electron (okwaziwa ngokuthi "i-oxidation reaction") kanye nokwakheka kwama-ion aqondile (ama-molecule ashajiwe). |
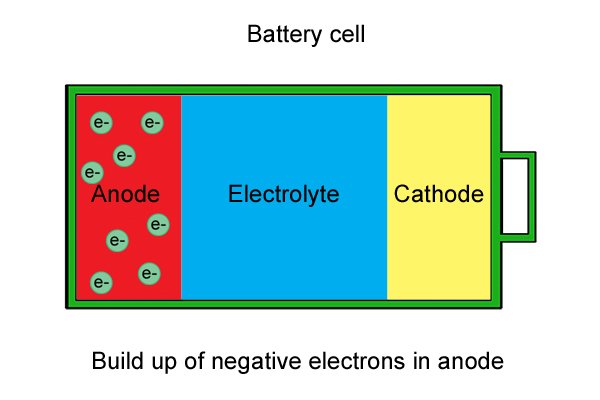
 | Lawa ma-electron "amahhala" anqwabelana ku-anode, ayenze ibe negethivu. |
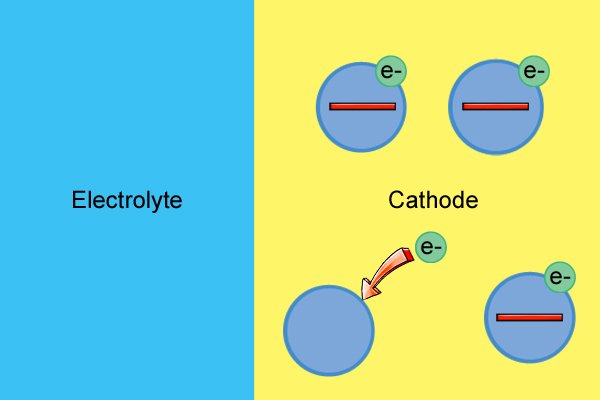
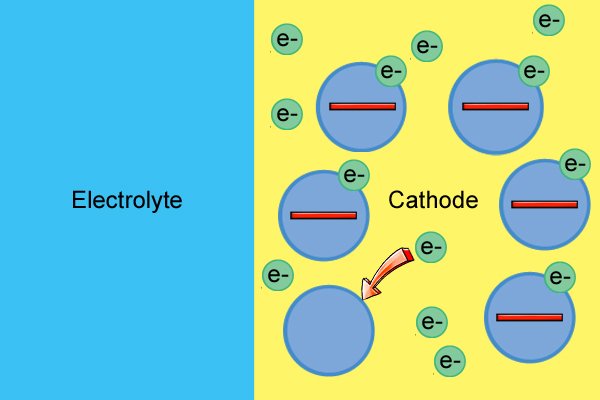
 | Ama-molecule e-cathode nawo awathathi hlangothi kuze kube yilapho esabela nge-electrolyte, esebenzisa ama-electron amahhala ukuze enze ama-ion angalungile (aziwa ngokuthi ukusabela kokunciphisa). |
 | Ukusetshenziswa kwama-electron mahhala kubangela ukuba i-cathode ibe ngcono nakakhulu kuze kube yilapho engekho ama-electron asele. |
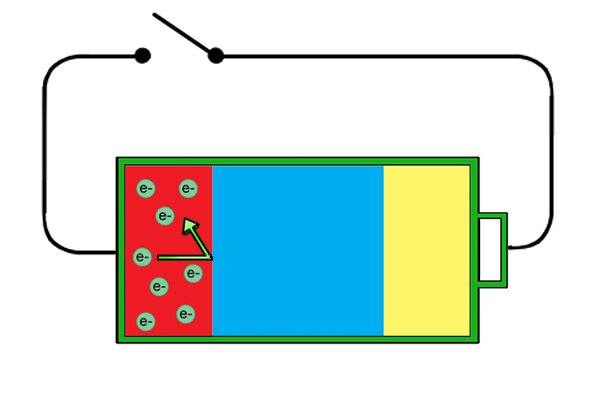
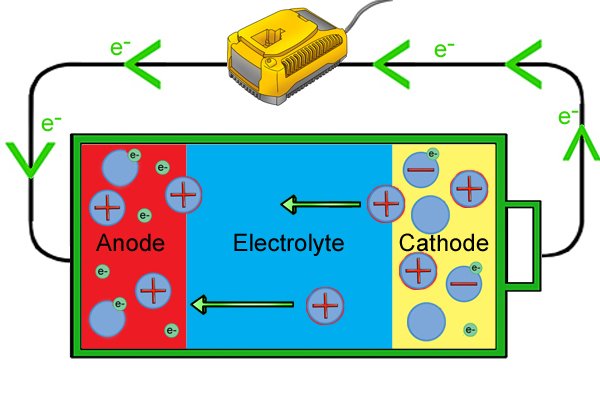
 | I-anode manje ixosha ama-electron futhi i-cathode iyawafuna, kodwa uma isifunda singaphelele, ama-electron amahhala ku-anode awakwazi ukuya ku-cathode ngoba awakwazi ukudlula ku-electrolyte. |
 | Lapho umjikelezo usuqediwe, ama-electron amahhala angageleza kumqhubi ukusuka ku-anode kuya ku-cathode. Njengoba bedlula ethuluzini, amandla abawaphetheyo angasetshenziswa ukwenza "umsebenzi", njengokuphendula umshini wokubhoboza okungenantambo. |
 | Lapho befika ku-cathode, bahlinzeka ngama-electron ukuze baqhubeke nokusabela kokunciphisa, bakhiqize ama-ion angalungile nakakhulu njengoba ama-electron engezwa. |
 | Phakathi naleso sikhathi, ku-anode, ukulahlekelwa kwama-electron kubangela ukwakheka kwama-ion amahle nakakhulu, akhangwa ama-ion angalungile ku-cathode, ngakho-ke ama-ion amahle aqala ukuhamba nge-electrolyte futhi axubane nama-ion angalungile ku-cathode. . |
 | Uma wonke ama-ion ephozithivu esethuthele ku-cathode futhi engasekho ama-electron amahhala asele, ibhethri liyayeka ukusebenza kahle futhi lidinga ukushajwa kabusha. |
 | Amashaja adlulisa i-voltage ephakeme kune-voltage yebhethri ngebhethri elichithiwe. Lokhu kubangela ukuthi ukusabela kubhethri kubuyele emuva. |
 | Okokufaka kukagesi kusuka kushaja kubangela ukuthi ama-electron aku-cathode abuyele kumjikelezo ku-anode. Njengoba i-anode iba yimbi kakhulu ngenxa yawo wonke ama-electron, ama-ion aqondile e-anode aqala ukushiya i-cathode futhi adlule ku-electrolyte abuyele ku-anode lapho ejoyina khona ama-electron amahhala futhi angathathi hlangothi futhi. |
 | Ibhethri selilungele ukuhamba futhi! |